Amicus Therapeutics (FOLD) are higher by more than 30% in mid-morning trading on Thursday, after the company announced that it will seek in the second half of this year accelerated U.S. approval for its Fabry disease therapy — Migalastat– following a productive meeting with the FDA. If approved, Migalastat will be the first oral therapy for Fabry disease, a rare but serious congenital condition. Fabry disease affects about 10,000 people worldwide.
Amicus also said that it will seek European regulatory approval for the drug in the second quarter of this year, also ahead of expectations.
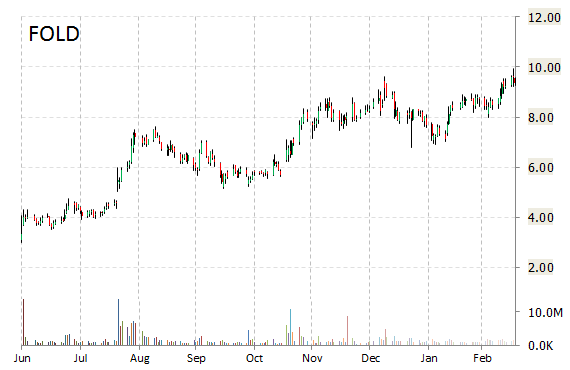
FOLD shares recently gained $2.93 to $12.28, its highest point since Feb. 6, 2009. The stock is up more than 308% year-over-year and has gained roughly 12.38% year-to-date. In the past 52 weeks, shares of Cranbury, New Jersey-based biopharmaceutical company have traded between a low of $1.77 and a high of $12.64.
Amicus Therapeutics, Inc. closed Wednesday at $9.35. The name has a total market cap of $1.17 billion.
Disclaimer: This page contains affiliate links. If you choose to make a purchase after clicking a link, we may receive a commission at no additional cost to you. Thank you for your support!


Leave a Reply