Shares of Depomed Inc. (DEPO) spiked 37% early Monday after the company announced U.S. regulatory approval of its pain medication Gralise. The drugmaker said the U.S. Food and Drug Administration approved Gralise, a once – daily treatment for pain following healing of the rash associated with shingles, triggering a milestone payment of $48 million from partner Abbott Laboratories (ABT). Depomed has licensed Gralise to Abbott Products, a unit of Abbott Laboratories.
The licensing deal with Abbott also includes sales milestone payments of up to $300 million, and calls for the Menlo Park, Calif. – based Depomed to receive royalties of up to 20% on sales of Gralise.
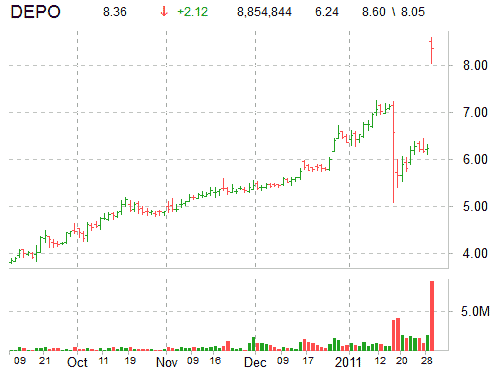
Shares of Depomed Inc are currently up 34.46% at $8.39 per share. The stock broke out to the upside earlier, rallying as high as 44%. Volume has exploded with more than 8.8 million DEPO shares already trading hands compared to a daily average volume of just 900,000 shares.
Depomed’s stock opened at 8.50 on Monday. The day’s trading range for the ticker, which currently has a median Wall Street price target of $8.00 with a high target of $9.00, has been between $8.05 and $8.60 per share.
Depomed has a market cap of $442 million and a P/E of 10.30.


Leave a Reply