
Apple (AAPL) seems ready to go big with the iPad as sales of the tablet are facing slowing growth for the first time since its 2010 debut.
According to 9to5Mac’s Mark Gurman, Cupertino plans to launch two new 12-inch iPads, introduce dual-app viewing mode, and add multi-user support to its iOS mobile OS. The publication said the jumbo-sized iPads, internally codenamed “J98″ and “J99, would arrive later this year, while support for dual apps could be introduced as early as next month.
Shares of Apple closed at $131.39 on Thursday.
—
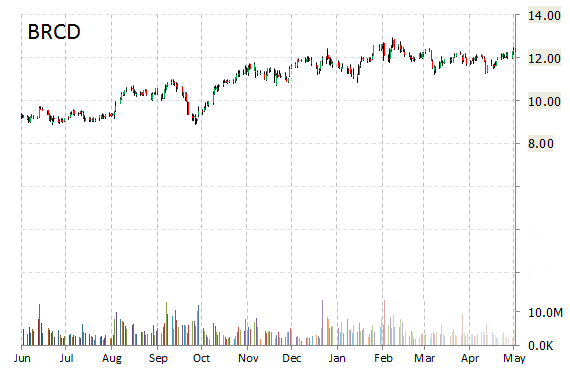
Brocade Communications Systems, Inc. (BRCD) shares are down $0.58 to $11.44 in after-hours trading Thursday after the company reported its second quarter earnings results.
The networking solutions firm posted earnings of $0.22 per share on revenues of $547 million, up 1.9% from $537 million a year ago. Analysts were expecting EPS of $0.20 on revenues of $551.33 million. Net income for the period ended May 2, 2015 came in at $77 million, or $0.18 per diluted share, from a loss of $14 million, or ($0.03) per diluted share, a year earlier.
“We significantly grew our IP Networking revenue year over year and closed the acquisitions of Connectem and the SteelApp assets, expanding our strong portfolio of virtual IP networking services,” said in a statement Lloyd Carney, CEO of Brocade. “These next-gen software-based technologies, together with our hardware products, allow us to build more strategic solutions for our customers and underscore our vision for the New IP.”
Looking forward, Brocade guided Q3/15 revenues of $540 – $560 million, as compared to analysts’ expectations of $561.36 million. The management also gave its bottom line range of $0.21 – $0.23 per share, against projections of $0.22 per share.
Cash Position: The company’s liquidity segment experienced a marginal increase during 2Q/15 to $1.37 billion from $1.25 billion during the same period last year. The $5.08 billion market cap firm reported $1.47 billion in debt in its most recent quarter.
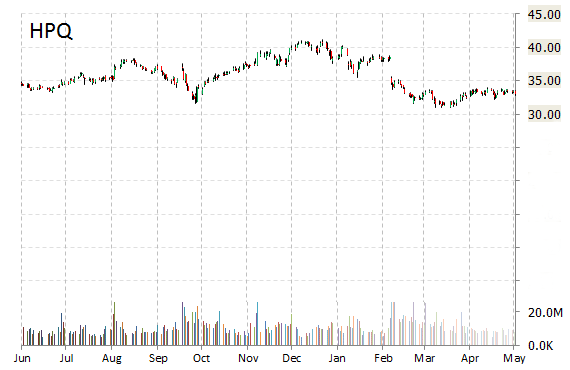
Hewlett-Packard Company (HPQ) reported second quarter non-GAAP EPS of $0.87 after the closing bell Thursday, compared to the consensus estimate of $0.86. Revenues decreased 6.8% from last year to $25.45 billion. Analysts expected revenues of $25.67 billion. Net income for the period ended April 30, 2015 was $1.01 billion, or $0.55 per share on a diluted basis, as compared to $1.27 billion, or $0.73 per share on a diluted basis, for the first quarter of 2014.
“Despite some tough challenges, we executed well across many parts of our portfolio, sustained our commitment to innovation, and delivered the results we said we would,” HP CEO Meg Whitman said in the company’s earnings release. “HP is becoming stronger as we head into the second half of our fiscal year and separation in November.”
For the current quarter, the PC maker said it expects EPS of $0.83 – $0.87 versus consensus of $0.87 per share on revenue of $25.87 billion. For full fiscal year 2015 which ends in October, HP expects earnings of $3.53 – $3.73 a share, compared to analysts’ estimates of $3.64 a share.
Cash – As of April 30, 2015, the $61.49 billion market cap company had cash, cash equivalents and short-term investments of $14.7 billion, compared to $15.3 billion as of October 31, 2014.
The stock is currently up $0.42 to $34.25 on 13.10 million shares.
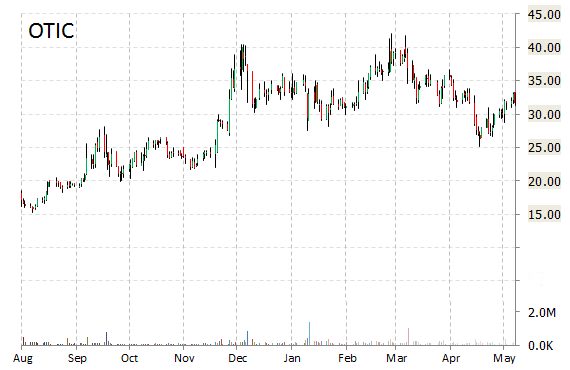
Shares of Otonomy, Inc. (OTIC) are lower by nearly 17% to $25.76 in after-hours trading on Thursday after the clinical-stage biopharmaceutical company announced that its Phase 2b Topline Data for OTO-104 in Meniere’s Disease, narrowly missed primary endpoint of reduction in vertigo frequency during Month 3.
“While the trial narrowly missed achieving statistical significance on the primary endpoint, we believe the clear efficacy trends observed along with the multiple secondary endpoints where statistical significance was achieved provide a solid basis to move OTO-104 forward into Phase 3 testing,” stated David A. Weber, Ph.D., president and CEO of Otonomy. “We plan to request an End-of-Phase 2 meeting with the FDA and intend to initiate two parallel Phase 3 trials in Meniere’s disease with the first beginning by the end of 2015.”
Disclaimer: This page contains affiliate links. If you choose to make a purchase after clicking a link, we may receive a commission at no additional cost to you. Thank you for your support!


Leave a Reply