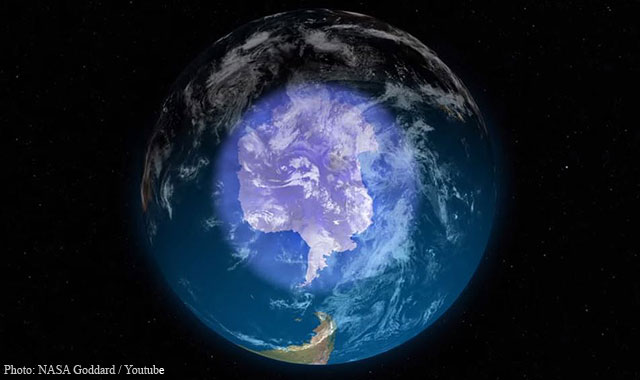
NASA and NOAA (the National Oceanic and Atmospheric Administration) have been measuring for more than thirty years now the size of the “hole” in Earth’s ozone layer – which forms over Antarctica every year. And every year, this specific layer of the atmosphere which protects all life on our planet reaches a maximum peak at the end of the southern hemisphere winter, a phenomenon known as the “annual maximum”.
On September 11, the hole’s annual peak extent was measured at 7.6 million square miles (sm) wide, marking the smallest record since 1988. 2017’s annual maximum, an area about 2.5 times the size of the US, was less than the peaks recorded for 2016 (8.9 million sm) and 2015 (10.9 million sm). To put things into perspective, the largest hole ever reported was in year 2000 with a peak extent of 11.5 million square miles wide.
Since 1991, the hole’s average annual maximum has been 10 million sm. Looking through these numbers alone, the continuous shrinkage appears to be a hopeful sign that the ozone layer has already started to heal and recover.
Surprisingly, scientists have attributed the decline more to warmer-than-average stratospheric weather conditions than to human intervention. Antarctic’s lower stratosphere has been warmer and more unstable since 2016, an odd whether pattern which has minimized the formation of stratospheric clouds. With fewer clouds, there are fewer sites for ozone-depleting chemical reactions to take a foothold. That said however, while warmer weather conditions have reduced ozone depletion for the last two years, the hole is still large compared to its size in the 1980s.
The ozone (chemical symbol: O3) is a gaseous molecule made of three oxygen atoms. At the surface level, ozone is toxic, but 7 to 25 miles high in the atmosphere, ozone forms a critical layer that protects the Earth’s surface from the harmful effects of the Sun’s ultraviolet radiation (UV rays). UV rays can cause sunburn, skin cancer, cataracts and skin and are lethal to living things.
1985 was the year when the massive ozone hole was first discovered. Scientists realized at the time that a class of compounds called chlorofluorocarbons (CFCs) were responsible for causing the amount of its depletion. The science behind the destruction was unveiled by Paul Crutzen, Mario Molina, and Frank Sherwood Rowland, a discovery that ultimately won them the 1995 Nobel Prize in Chemistry. CFCs are components used in spray cans, refrigerators, and air conditioners. After managing to reach the atmosphere, these components release chemical elements known as ‘chlorine or bromine radicals’. Once released, these highly halogen reactive elements – which it should be noted, in the ’80s were seen as a safer replacement for earlier industrial chemicals since they were non-toxic to humans – cause a chain reaction by initially breaking down ozone molecules to eventually progress toward depleting an entire ozone layer.
While the recent shrinkage of the ozone hole has been credited to natural variability, the fact should not in anyway minimize the efforts undertaken by concerned parties under the Montreal Protocol. On the other hand, the reality is that curbing ozone depletion is not achievable in the immediate term as complete removal of CFCs in our atmosphere are projected to take between 50 to 100 years. If the Montreal Protocol implementation remains on track, the earliest estimated time of bringing the ozone hole back to its 1980 levels is by year 2070. This estimate however, does not include the fact that back in June scientists identified Dichloromethane — a solvent widely used in various industrial processes with the power to destroy ozone – as a new possible threat to the ozone’s recovery process (we can’t seem to do without what we do best as humans, mess our planet…hopefully, we will change our ways).
This year marks the 30th anniversary of the original signing of the Montreal Protocol. Formally known as the “Montreal Protocol on Substances that Deplete the Ozone Layer”, it is an environmental treaty made to regulate and eventually phase out the use of CFCs and other ozone-depleting substances. It started only with 46 nations as signatories but today, it has grown to 197 parties.
Disclaimer: This page contains affiliate links. If you choose to make a purchase after clicking a link, we may receive a commission at no additional cost to you. Thank you for your support!




There is absolutely zero proof the ozone “hole” was ever man made, nor is there proof that any conservation or environmental effort are the cause of its shrinkage. What we have here is speculation and conjecture, nothing more…
Ditto that. I did not believe them then, nor do I now