
BioCryst Pharmaceuticals (BCRX) shares rose as much as 7%, to reach a 52 week high of $13.65 on Monday, as a positive article by Forbes’ Paul Rodgers on the company’s Ebola drug makes the rounds.
According to Rodgers, who cites the respected scientific journal ‘Nature’, BioCryst’s new drug BCX4430 has shown 90% efficacy in animal models by blocking the reproduction of the Ebola virus inside infected cells. The drug has also demonstrated a broad-spectrum activity in multiple viruses and a favorable preclinical safety profile.
BioCryst scientists, who are developing BCX4430 in collaboration with U.S. Government Agencies, are optimistic that the drug will prove effective against filoviruses such as Ebola virus and Marburg virus, which are extremely virulent and are classified as Category A Bioterrorism Agents by the Centers for Disease Control and Prevention, because it is a “wide spectrum” agent, with evidence that it could also work against SARS and Yellow Fever.
“We’re going absolutely as fast as we can,” BioCryst’s Dr Bill Sheridan, co-leader of the research team for BCX4430 told Rodgers. “If we replicate what we’ve seen so far, there’s a pretty good shot of the drug being helpful in humans.”
BCX4430 was first synthesised by the biotech company at its lab in Birmingham, Alabama, during research into treatments for Hepatitis C.
BioCryst Pharmaceuticals shares are currently trading at an unusually high volume with more than 2.5 million shares changing hands at $13.29 as of 1:45 pm ET, well ahead of its three month daily average of 1.2 million shares.
In the past 52 weeks, shares of BioCryst Pharmaceuticals have traded between a low of $4.55 and a high of $ 13.65. The stock’s 50-day moving average is $12.00 and its 200-day moving average is $10.54.
The chart below shows where the equity has traded over the last 52 weeks, with the 50-day and 200-day MAs included.
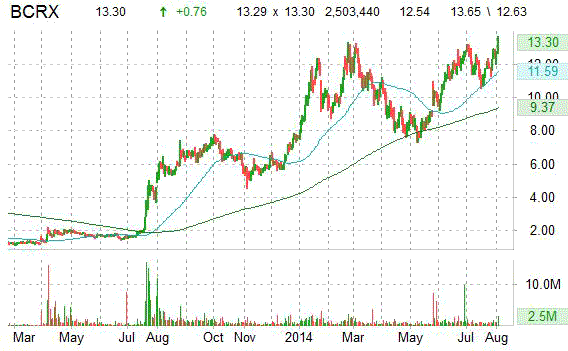
Over the past year ticker has gained 128% vs. 75.53% year-to-date.
Disclaimer: This page contains affiliate links. If you choose to make a purchase after clicking a link, we may receive a commission at no additional cost to you. Thank you for your support!


Leave a Reply